Toward Understanding pH
Let's begin with the words acidic and basic as extremes which describe solutions as hot and cold are extremes which describe temperature. Just as mixing hot and cold water evens out the temperature, mixing acids and bases can cancel their extreme effects and is then considered neutral.
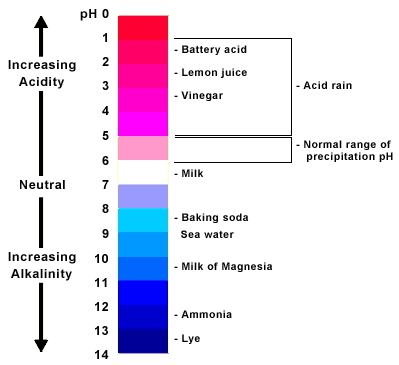
The pH scale can tell if a liquid is more acid or more base, just as the Fahrenheit or Celsius scale is used to measure temperature. The range of the pH scale is from 0 to 14 from very acidic to very basic. A pH of 7 is neutral. A pH less than 7 is acidic and greater than 7 is basic.
Each whole pH value below 7 is ten times more acidic than the next higher value. For example, a pH of 4 is ten times more acidic than a pH of 5 and a hundred times (10 X 10) more acidic than a pH of 6. This holds true for pH values above 7, each of which is ten times more basic (also called alkaline) than the next lower whole value. An example would be, a pH of 10 is ten times more alkaline than a pH of 9.
The pH Scale...
Acid-Base Indicators...
What is an acid-base indicator?
An acid-base indicator is a weak acid or a weak base. Indicators have a very useful property - they change color depending on the pH of the solution they are in. This color change is not at a fixed pH, but rather, it occurs gradually over a range of pH values. This range is termed the color change interval. Each pH indicator is defined by a useful pH range. For example Phenolphthalein changes from colorless at 8.0 to pink at 10.0. And Bromthymol Blue has a useful range from 6.0 (yellow) to 7.6 (blue).
See our more complete chart of common pH indicators with ascending pH ranges.
How are liquid indicators used?
Liquid pH indicators are used to test other solutions. A few drops of the right indicator added to an unknown solution can tell you its pH value. Chemists use pH indicators in a common laboratory procedure called titration. Here, an unknown substance is measured by carefully adding a solution of known concentration until a neutral point is reached. The neutral point is indicated by the color change of a pH indicator mixed in with the unknown solution.
How else can pH be measured?
With test papers - In addition to liquid indicators, there are commercial calibrated test papers which are impregnated with pH indicators. pH is determined by immersing the strip in the liquid to be tested and comparing its color with a standard color chart provided with the pH paper.
With digital readout via electronic meter - In recent years, the demand for measuring pH has grown dramatically. This includes environmental, agricultural, wastewater, pharmaceutical, and educational applications, to name just a few.
Electronic, benchtop meters are available that read pH to resolutions of 0.001. Portable, "pocket size" meters suitable for a wide array of testing needs are now offered by The Science Company with accuracies of 0.1 or 0.01 pH. These easy-to-use meters feature built-in memorized buffer values for quick, "automatic" calibration and automatic temperature compensation which eliminates errors in pH measurement caused by solution temperature variations.
Electronic pH meters require periodic calibration using standard value calibration solutions (sometimes called buffer solutions). Typical standards are pH 4.01, pH 7.01 and pH 10.01.
Common Acid-base Indicators
- Indicator / Cat. No. / pH Range and Color
- Phenolphthalein / NC-0700 / 8.0 (colorless) to 10.0 (pink)
- Bromothymol Blue / NC-1949 / 6.0 (yellow) to 7.6 (blue)
- o-Cresolphthalein / NC-13358 / 8.2 (colorless) - 9.8 (violet)
- Bromophenol Blue / NC-13318 / 3.0 (yellow) to 4.6 (blue)
- Bromocresol Green / NC-13321 / 3.8 (yellow) - 5.4 (blue)
Commercial Calibrated Test Papers
- Test paper in 15 ft. roll. NC-0692
- 100 test strips in 4 colors. NC-0690


